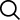[av_textblock size=” font_color=” color=” av-medium-font-size=” av-small-font-size=” av-mini-font-size=” av_uid=’av-jxxb0xl3′ admin_preview_bg=”]
Sulfuric Acid has an Important Role in Treating Wastewater, Making Lead Acid Batteries, Manufacturing Rayon, and Harvesting Potatoes as a Desiccant
[/av_textblock]
[av_hr class=’invisible’ height=’35’ shadow=’no-shadow’ position=’center’ custom_border=’av-border-thin’ custom_width=’50px’ custom_border_color=” custom_margin_top=’30px’ custom_margin_bottom=’30px’ icon_select=’yes’ custom_icon_color=” icon=’ue808′ font=’entypo-fontello’ av_uid=’av-rzogef’ admin_preview_bg=”]
[av_textblock size=” font_color=” color=” av-medium-font-size=” av-small-font-size=” av-mini-font-size=” av_uid=’av-jxyrv0du’ admin_preview_bg=”]
Wastewater Treatment
Wastewater treatment is a very important process that must be done in order to protect the people and animals living in our environment. As the most commonly used acid around the world, sulfuric acid is often used to bring the pH level of wastewater back to normal.
The major benefits of using sulfuric acid to treat wastewater are as follows:
- Solid particles with chemicals such as ferric chloride, polymers, and alums will be eliminated.
- The wastewater will be brought back to a neutral pH level, which is a process known as neutralization.
- Offensive odors found in wastewater will be controlled and prevented.
- Pathogens that can spread diseases are destroyed through disinfection.
- Water and sludge are separated through chemical treatment which makes it easier to remove the sludge.
[/av_textblock]
[av_image src=’https://ecolink.com/wp-content/uploads/2019/07/wastewater-treatment-2-300×251.jpg’ attachment=’14382′ attachment_size=’medium’ align=’center’ styling=” hover=” link=” target=” caption=” font_size=” appearance=” overlay_opacity=’0.4′ overlay_color=’#000000′ overlay_text_color=’#ffffff’ copyright=” animation=’no-animation’ av_uid=’av-jxyrwa4n’ admin_preview_bg=”][/av_image]
[av_hr class=’invisible’ height=’35’ shadow=’no-shadow’ position=’center’ custom_border=’av-border-thin’ custom_width=’50px’ custom_border_color=” custom_margin_top=’30px’ custom_margin_bottom=’30px’ icon_select=’yes’ custom_icon_color=” icon=’ue808′ font=’entypo-fontello’ av_uid=’av-rzogef’ admin_preview_bg=”]
[av_textblock size=” font_color=” color=” av-medium-font-size=” av-small-font-size=” av-mini-font-size=” av_uid=’av-34fjgw7′ admin_preview_bg=”]
Making Lead Acid Batteries
In its diluted form, sulfuric acid acts as an electrolyte to encourage the flow of electrons between plates in a battery. When sulfuric acid is used to perform this task, it is known as battery acid. Sulfuric acid is consumed during the discharge process, but during the charge process, sulfuric acid is released into the electrolyte because lead sulphate is converted back to lead and lead dioxide. As a result, the specific gravity of the electrolyte is used to determine to the state of charge.
An electrolyte of sulfuric acid is used during these basic reactions due to the uptake and release of sulfuric acid molecules. Its strength usually varies between 28 to 32 percent or between 4.2 to 5 Molar, according to the battery manufacturer. Invented in 1859, these sealed-unit lead acid batteries are mostly used in the automotive industry for trucks and cars.
[/av_textblock]
[av_image src=’https://ecolink.com/wp-content/uploads/2019/07/lead-acid-batteries-300×251.jpg’ attachment=’14383′ attachment_size=’medium’ align=’center’ styling=” hover=” link=” target=” caption=” font_size=” appearance=” overlay_opacity=’0.4′ overlay_color=’#000000′ overlay_text_color=’#ffffff’ copyright=” animation=’no-animation’ av_uid=’av-2r0oxaf’ admin_preview_bg=”][/av_image]
[av_hr class=’invisible’ height=’35’ shadow=’no-shadow’ position=’center’ custom_border=’av-border-thin’ custom_width=’50px’ custom_border_color=” custom_margin_top=’30px’ custom_margin_bottom=’30px’ icon_select=’yes’ custom_icon_color=” icon=’ue808′ font=’entypo-fontello’ av_uid=’av-rzogef’ admin_preview_bg=”]
[av_textblock size=” font_color=” color=” av-medium-font-size=” av-small-font-size=” av-mini-font-size=” av_uid=’av-27eeecn’ admin_preview_bg=”]
Manufacturing Rayon
Rayon is referred to as artificial silk and considered to be a steady semi-synthetic textile. It is a soft, smooth, and cool fabric that can easily be dyed, but unlike silk, Rayon is perfect to use in countries with hot and humid weather because it does not insulate body heat.
To make textile Rayon, cellulose fibers derived from wood are dissolved in a Tetra Amine Copper (II) solution that produces a thick blue liquid. This thick blue liquid is then injected into sulfuric acid which results in Rayon fibers. After awhile, the copper salts from the fibers wash out and the fibers will become colorless, due to a reaction between sulfuric acid and the complex compound.
[/av_textblock]
[av_image src=’https://ecolink.com/wp-content/uploads/2019/07/rayon-1-300×251.jpg’ attachment=’14384′ attachment_size=’medium’ align=’center’ styling=” hover=” link=” target=” caption=” font_size=” appearance=” overlay_opacity=’0.4′ overlay_color=’#000000′ overlay_text_color=’#ffffff’ copyright=” animation=’no-animation’ av_uid=’av-jxxg6n5c’ admin_preview_bg=”][/av_image]
[av_hr class=’invisible’ height=’35’ shadow=’no-shadow’ position=’center’ custom_border=’av-border-thin’ custom_width=’50px’ custom_border_color=” custom_margin_top=’30px’ custom_margin_bottom=’30px’ icon_select=’yes’ custom_icon_color=” icon=’ue808′ font=’entypo-fontello’ av_uid=’av-rzogef’ admin_preview_bg=”]
[av_textblock size=” font_color=” color=” av-medium-font-size=” av-small-font-size=” av-mini-font-size=” av_uid=’av-3un38ef’ admin_preview_bg=”]
Harvesting Potatoes
In order to prevent entanglement in harvesting equipment, the stem of the potato must be dried out. By spraying the potato fields with a sulfuric acid solution, potato farmers ensure that the green tops will shrink back and blacken within a day or two. Potato farmers hire special contractors to carry out this task.
The advantages of using sulfuric acid, instead of another common desiccant, are its speedy kill on contact and capability to eliminate pathogen sores at the surface of the soil, such as Phytophthora infestans. However, pathogens under the debris that are not in direct contact with the sulfuric acid will not be affected. The vine kill is so fast though, that the tubers cannot mature or set skin.
[/av_textblock]
[av_image src=’https://ecolink.com/wp-content/uploads/2019/07/potatoes-300×251.jpg’ attachment=’14385′ attachment_size=’medium’ align=’center’ styling=” hover=” link=” target=” caption=” font_size=” appearance=” overlay_opacity=’0.4′ overlay_color=’#000000′ overlay_text_color=’#ffffff’ copyright=” animation=’no-animation’ av_uid=’av-c2mqnb’ admin_preview_bg=”][/av_image]
[av_textblock size=” font_color=” color=” av-medium-font-size=” av-small-font-size=” av-mini-font-size=” av_uid=’av-jxyp45ws’ admin_preview_bg=”]
If you would like to purchase sulfuric acid, contact Ecolink at (800)-563-1305 or email info@ecolink.com
If you have any further questions, submit an inquiry through our online form.
[/av_textblock]